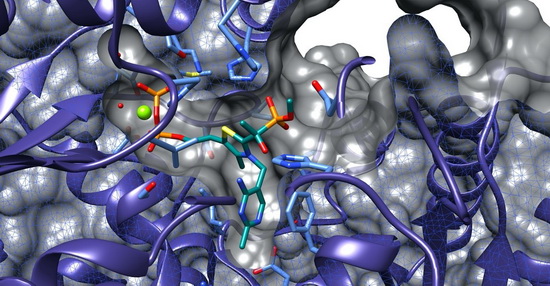
 The project will be focussed on the elucidation of crystal structures of constituent enzymes of the MEP pathway from pathogenic organisms (Pseudomonas aeruginosa, Plasmodium falciparum, Mycobacterium tuberculosis, Escherichia coli).
The project will be focussed on the elucidation of crystal structures of constituent enzymes of the MEP pathway from pathogenic organisms (Pseudomonas aeruginosa, Plasmodium falciparum, Mycobacterium tuberculosis, Escherichia coli).
Over the past decades, structural biologists have extensively studied the MEP pathway enzymes. Nevertheless, important gaps remain to be addressed. By adopting an extensive structural-genomics approach for each constituent enzyme, we will be able to shed light on the structural features of the enzymes across a series of important pathogens.
The students will be trained to establish constructs and crystallization conditions and to carry out high-throughput screening of fragment libraries by X-ray crystallography.
ESR 1: Biophysical and structural characterisation of DXS, DXR and IspD (in complex with inhibitors)
The project aims to generate DXS construct for P. falciparum and P. aeruginosa organisms. Available M. tuberculosis DXS X-ray crystal will provide a springboard for the ESR in the initial period. In a similar manner, DXR and IspD enzymes will be tackled and, this time, the ESR will be supported by the Escherichia coli and M. tuberculosis IspD crystal structures. In a collaborative approach, ESR 4 and ESR 5 will pursue a structure-based drug discovery approach thanks to crystal structures of ligand–enzyme complexes.
Supervisor: Prof. Matthew Groves
Researcher: Victor Gawriljuk
Secondments: Commissariat à l’énergie atomique et aux énergies alternatives (CEA) /Academia Sense About Science (SAS) / Industry
ESR 2: Biophysical and structural characterisation of IspE and IspF (in complex with inhibitors)
Efficient recombinant protein-production methods and robust protocols for crystallisation targeting IspE and IspF from P. aeruginosa and P. falciparum will be developed by ESR 2. Hit compounds identified by ESR 6, ESR 8 and ESR 9 will be used to generate accurate co-crystal structures complex.
Supervisor: Prof. Wulf Blankenfeldt
Researcher: Camilla Ornago
Secondments: Commissariat à l’énergie atomique et aux énergies alternatives (CEA) /Academy BioSolveIT (BIT) / Industry
ESR 3: Biophysical and structural characterisation of IspG and IspH (in complex with inhibitors)
The last two enzymes of the MEP pathway (IspG and IspH) are metalloenzymes containing a highly oxygen-sensitive [4Fe-4S] cluster. Therefore, the production of the enzymes must be done in anaerobic conditions. In close collaboration with ESR 7 at UNISTRA, who is in charge of the synthesis of selective inhibitors for these two enzymes, ESR 3 will perform in silico docking and modelling studies.
Supervisor: Dr. Franck Borel
Researcher: Lucia D’Auria
Secondments: Université de Strasbourg (UNISTRA) / Academia Helmholtz-Zentrum für Infektionsforschung (HZI) / Academia Drug for Neglected Disease Initiative (DNDI)/ Industry
NEWS
- MepAnti Drug Research Symposium: Young Minds, Big ImpactAnnouncement: the MepAnti team is organizing the MepAnti Drug Research Symposium: Young Minds, Big ImpactMore information about the symposium can be found here /*! elementor …
Continue reading "MepAnti Drug Research Symposium: Young Minds, Big Impact"
FUNDING

This project has received funding from the European Union's Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie grant agreement No 860816.
